Polymerisation
Monomers
Organic compounds are typically composed of recurring subunits (monomers) which are covalently joined to form polymers
-
The monomeric subunit of carbohydrates is a monosaccharide (single sugar unit)
-
Nucleic acids are composed of repeating nucleotides (containing a sugar, phosphate and nitrogenous base)
-
Proteins consist of linked chains of amino acids which differ according to a variable side chain (‘R’ group)
-
Lipids do not contain monomers but certain types may be composed of distinct subunits (fatty acid chains)
Types of Monomers / Subunits
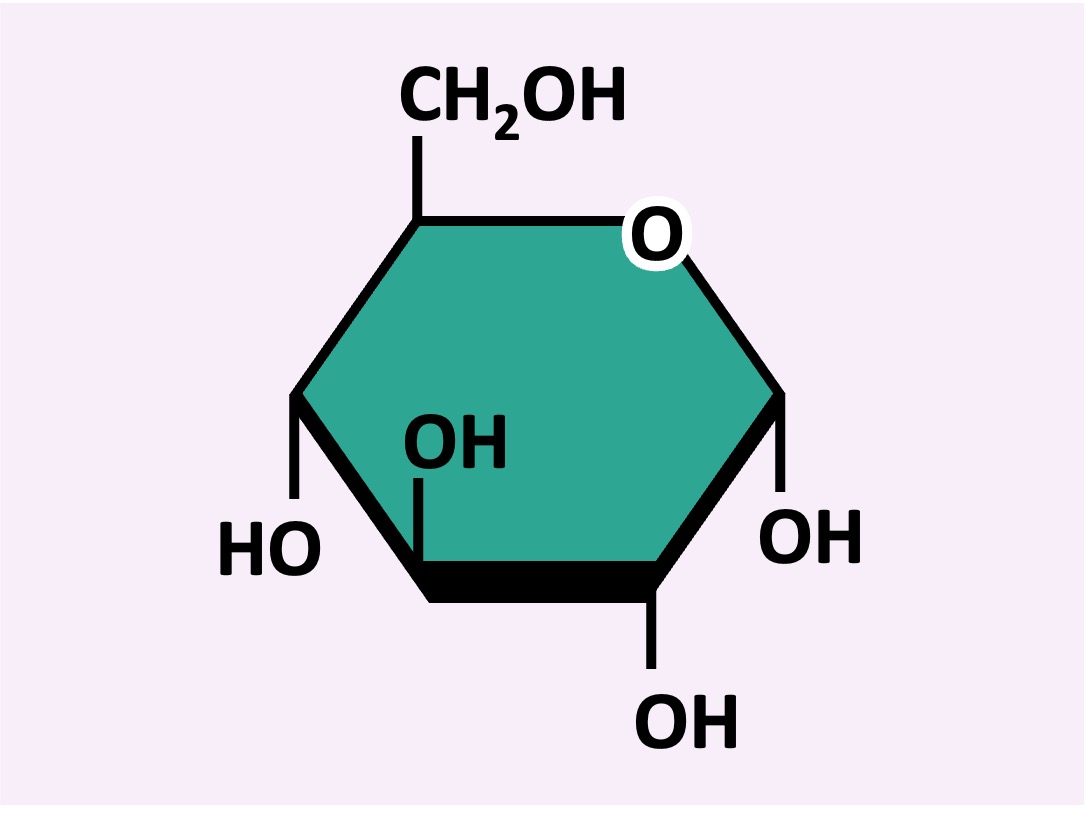
Monosaccharide
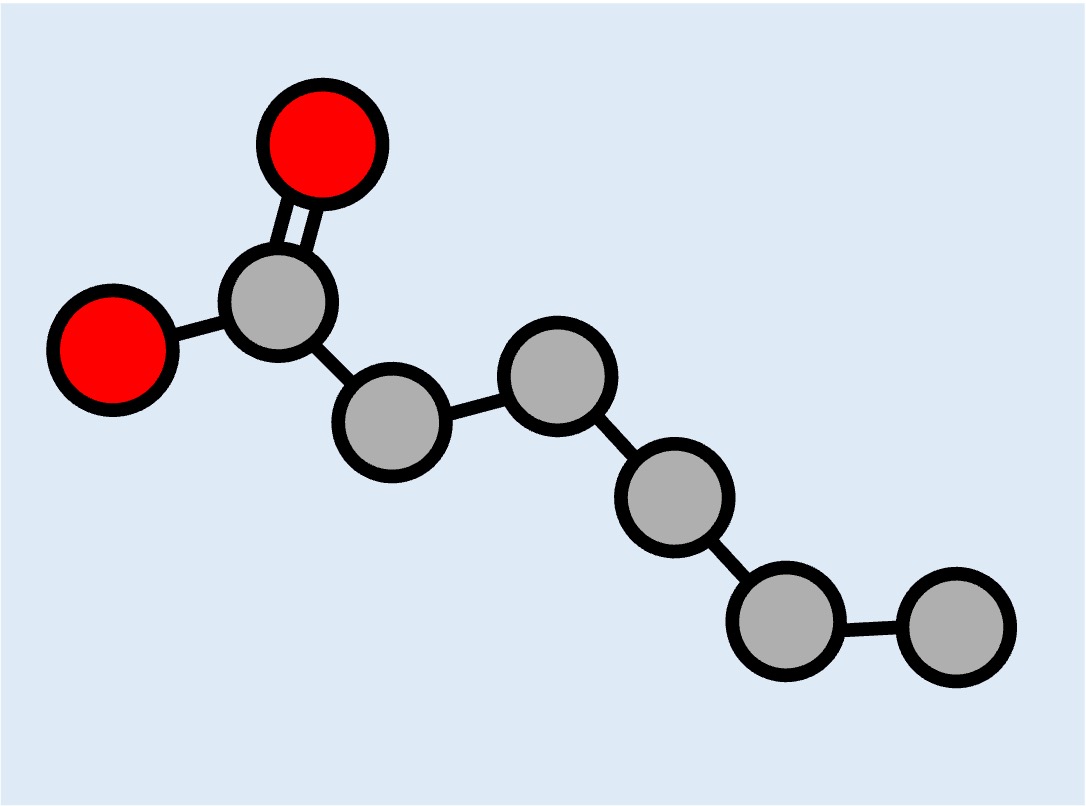
Fatty Acid
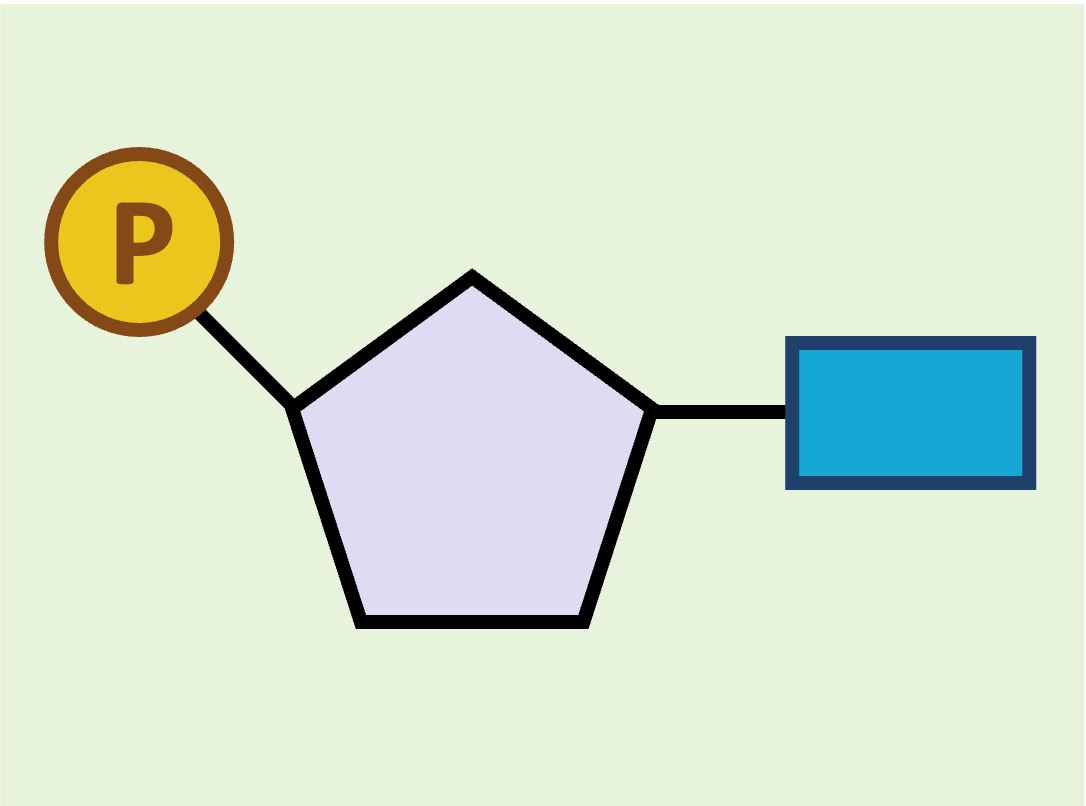
Nucleotide
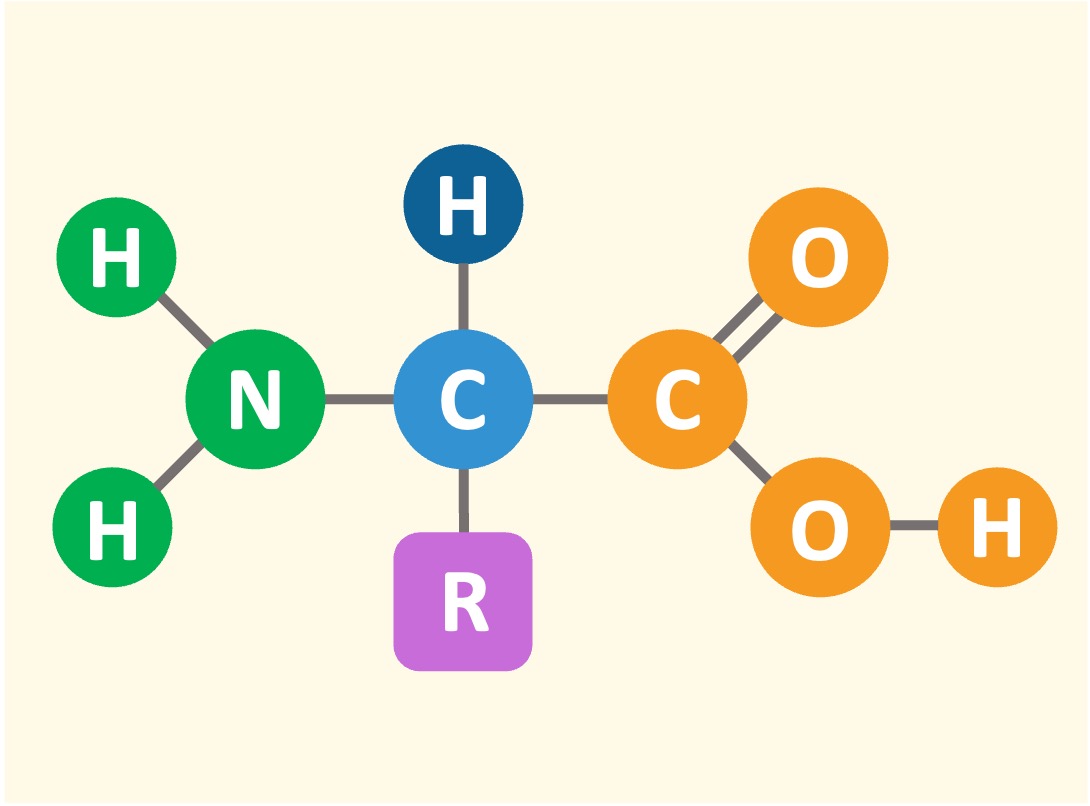
Amino Acid
Polymerisation
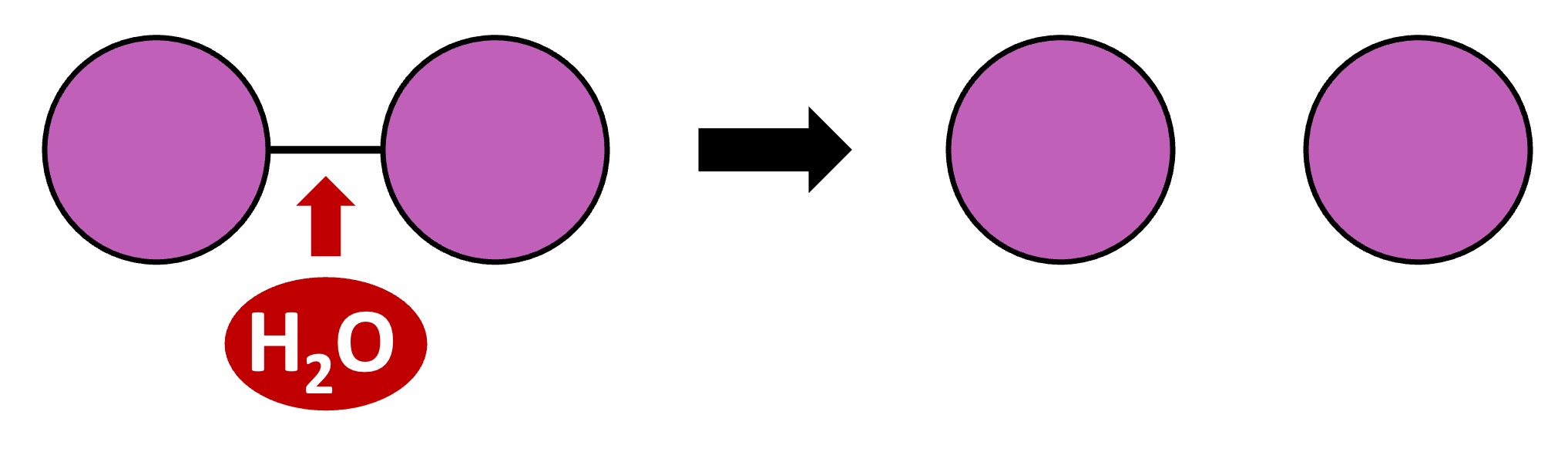
Polymers can be formed from monomeric subunits via condensation reactions
-
A hydroxyl group (-OH) on one monomer is combined with a hydrogen atom (-H) on another monomer
-
The two monomers become covalently bonded and a water molecule is produced as a by-product
Condensation Reaction
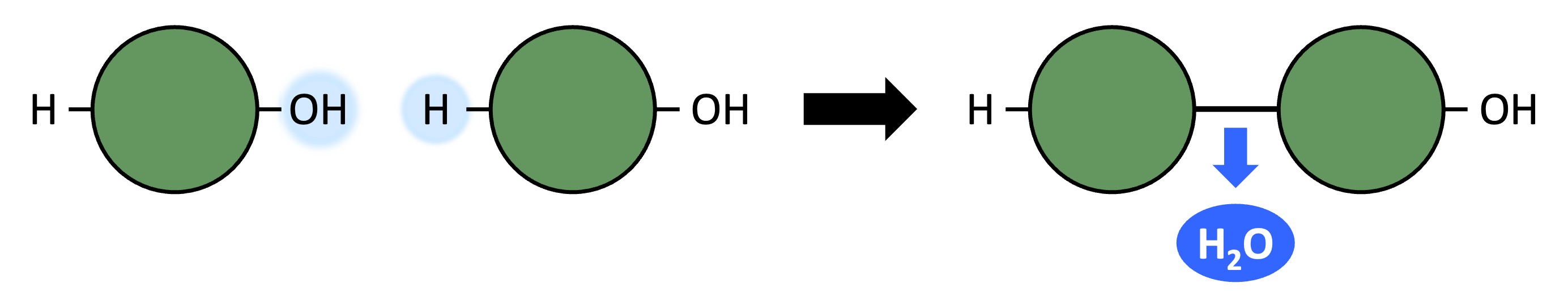
Condensation Reaction
Polymers
Polymers are large macromolecules composed of smaller repeating subunits (called monomers) via condensation reactions
-
Carbohydrates: Monosaccharides are joined together to form polysaccharides via glycosidic linkages
-
Nucleic Acids: Nucleotides are connected by phosphodiester bonds to form polynucleotide chains (DNA or RNA)
-
Proteins: Amino acids are linked by peptide bonds to form polypeptide chains (proteins can possess multiple chains)
-
Lipids: Do not possess monomers but fatty acid chains can be connected by ester linkages to form triglycerides and phospholipids
Types of Polymers
Carbohydrates
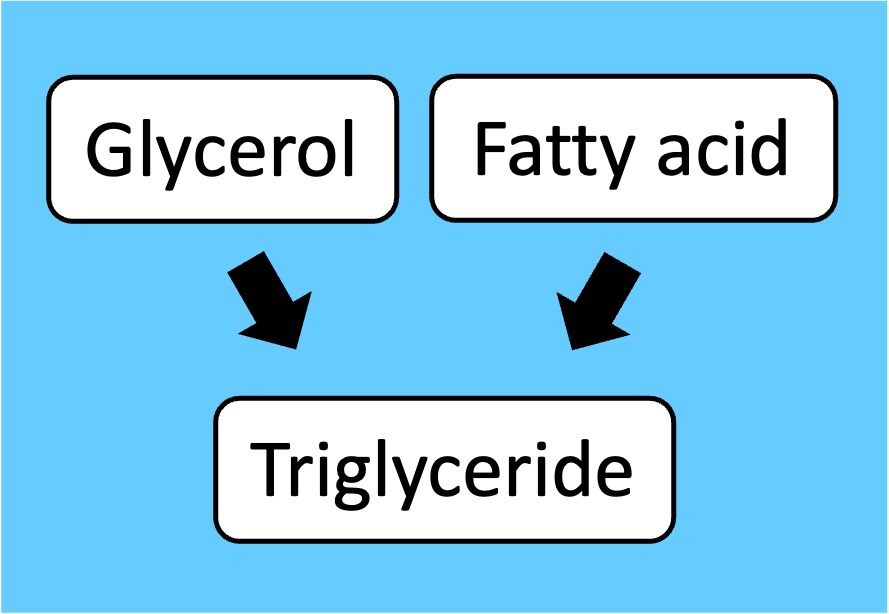
Lipids*
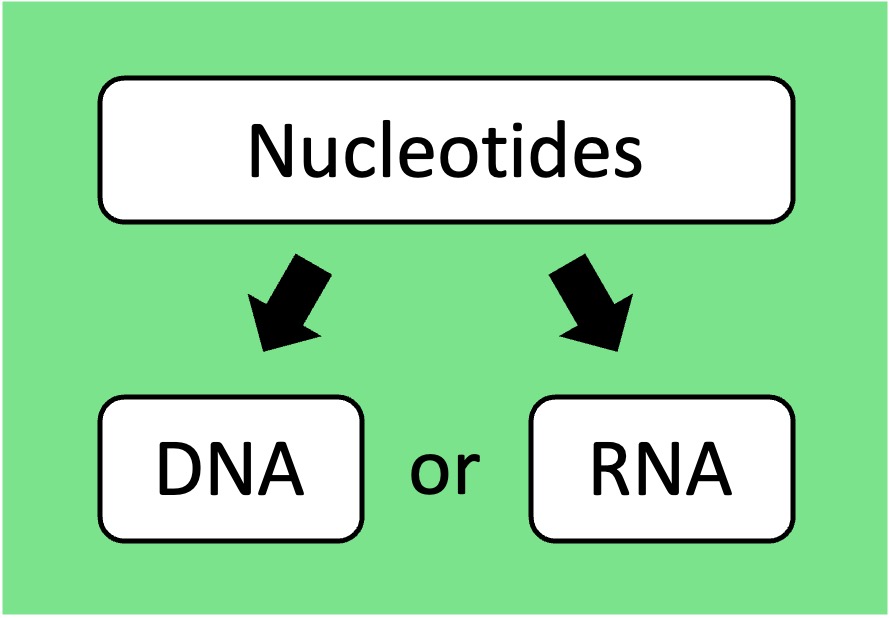
Nucleic Acids
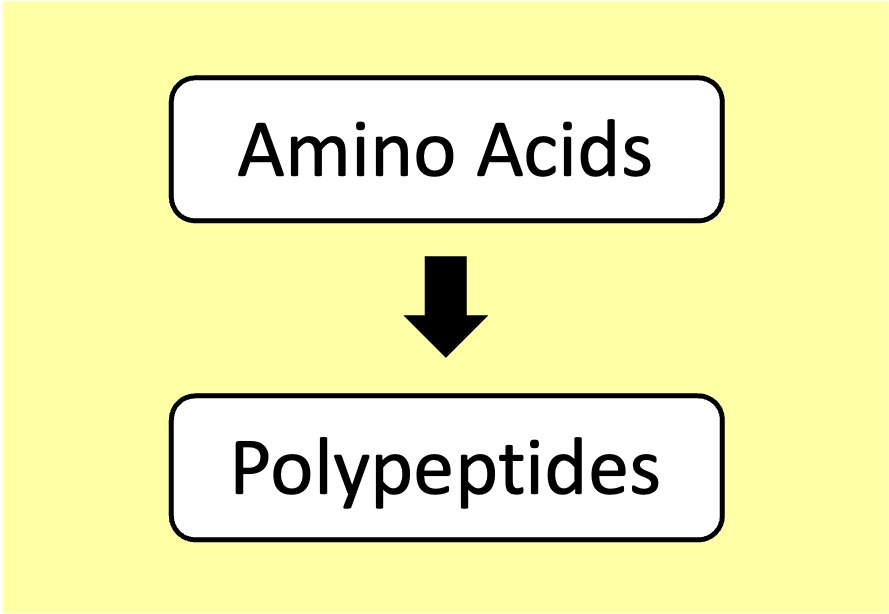
Proteins
Digestion
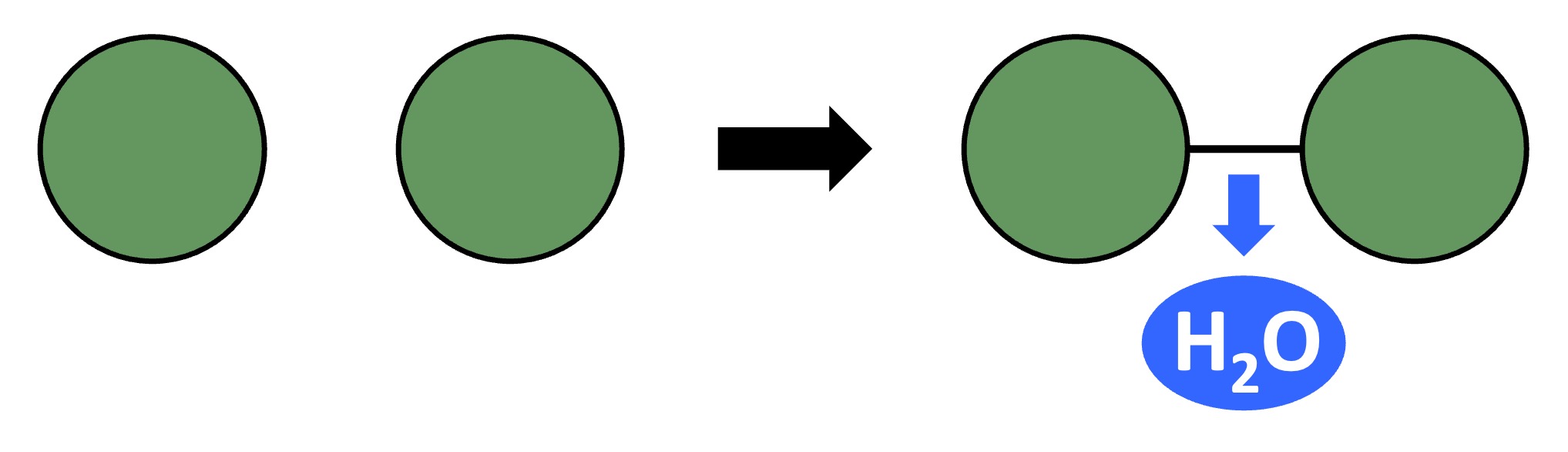
Polymers can be broken down into their monomeric subunits via hydrolysis reactions
-
A water molecule is split to provide the -H and -OH groups required to break the covalent bond between two monomers
Hydrolysis Reaction
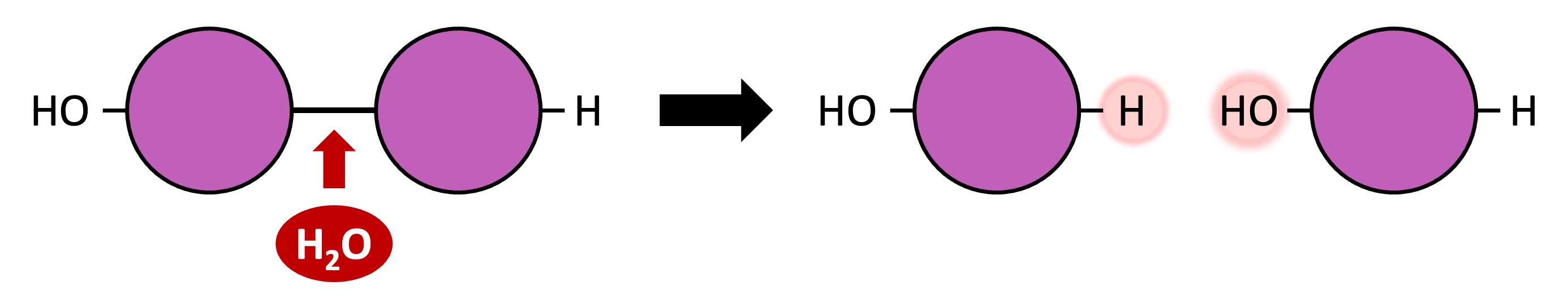
Hydrolysis Reaction